Abu Dhabi official who received COVID-19 inactivated vaccine ‘feeling fine’ after 24 hours
Dr Jamal Al Kaabi said he is excited about the trials, has “a good feeling” about them
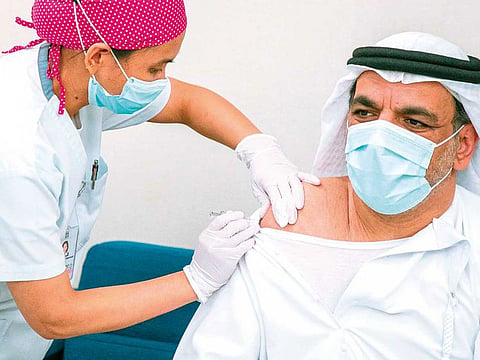
Abu Dhabi: The second person to be administered the coronavirus vaccine in trials in Abu Dhabi said he was “feeling fine” after 24 hours.
Dr Jamal Al Kaabi, acting undersecretary at Abu Dhabi healthcare regulator, the Department of Health (DoH), told Gulf News that he “excited to be part of this historic moment”.

“I did the tests and received the vaccine at 6pm on Wednesday. And I feel good, praise be to Allah. It was like a normal flu shot,” said Dr Al Kaabi, who is an Emirati physician by profession.
He was one of two senior officials who kicked off the Phase III vaccine trials in Abu Dhabi. The first was Sheikh Abdullah bin Mohammed Al Hamed, DoH chairman.
The inactivated vaccine has been developed by Chinese pharmaceutical giant, Sinopharm Chinese National Biotec Group, and has successfully and safely passed through the first two phases of clinical trials with no adverse effects reported. It has also allegedly produced antibodies in all participants who received two doses of the vaccine 28 days apart.
Abu Dhabi-based artificial intelligence and cloud computing firm, G42, has partnered with Sinopharm to sponsor the Phase III trials in the UAE. The trials will be conducted under the supervision of the UAE Ministry of Health and Prevention and the DoH, and by five public health facilities under Abu Dhabi public health provider, the Abu Dhabi Health Services Company.
“I am really excited about this trial as a physician. I have always believed in the importance of developing a research ecosystem, and personally supported research initiatives. Fortunately, we received support from our leadership for these trials,” Dr Al Kaabi said.
“It is the beginning of a historic moment, and God willing, we will have a great conclusion to these trial. I have a good feeling,” the official said.
Officials announced at a virtual press conference today (June 16) that recruitment for the trials will begin next week. The aim is to enroll 15,000 people, with 5,000 for the initial stages to be carried out in Abu Dhabi.







