Latest COVID-19 vaccine developments, all you need to know
AstraZeneca signs Japan, Chinese deals, Novavax signs Serum Institute of India
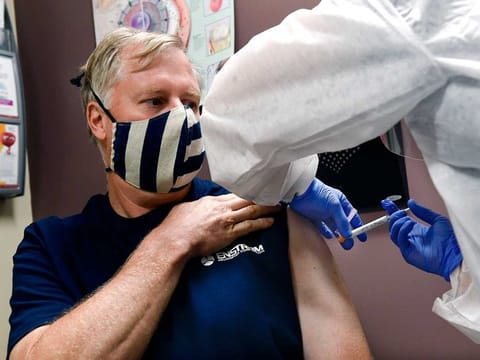
HIGHLIGHTS
- Japan in deals with AstraZeneca, Novavax for COVID-19 vaccines
- Novavax signs Covid-19 vaccine supply deal with India's Serum Institute of India
- AstraZeneca signs deal with China's Shenzhen Kangtai Biological Products to supply its COVID-19 vaccine candidate in mainland
Also In This Package
Japan's deal with AstraZeneca
TOKYO: The Japanese government has reached an agreement with British drugmaker AstraZeneca Plc to receive a supply of 120 million doses of a coronavirus vaccine being developed with the University of Oxford, health minister Katsunobu Kato said Friday.
Japan's Kyodo News reported that the vaccine will be supplied to Japan from next year if put into practical use, with 30 million doses to be received by March.
The drugmaker, which has been conducting a final-stage clinical trial of its experimental AZD1222 vaccine, has not yet decided whether it is necessary to inoculate a person once or twice.
AstraZeneca has announced that it will start supplying its COVID-19 vaccine overseas in September, and is expected to begin clinical trials in Japan later this month to confirm its efficacy and safety.
The British company is also in talks with JCR Pharmaceuticals Co. in Hyogo Prefecture to produce vaccine solutions, and with Tokyo-based Daiichi Sankyo Co. for packing and storing vaccines.
The drugmaker's COVID-19 vaccine is among the world's most advanced vaccine candidates. The company has said the vaccine showed robust immune responses in all participants in early-stage trials.
Japan's deal with Pfizer
Japan has already agreed to receive a supply of 120 million doses of a coronavirus vaccine for 60 million people by the end of June next year from U.S. pharmaceutical giant Pfizer Inc. and its German partner BioNTech SE, if they succeed in developing it.
Novavax signs vaccine supply deal with India's Serum Institute
Novavax Inc said on Wednesday it has entered a supply and license agreement with the Serum Institute of India for the development and commercialization of its COVID-19 vaccine candidate.
The Indian drugmaker will have exclusive rights for the vaccine in India during the term of the deal and non-exclusive rights during the "Pandemic Period" in all countries other than those designated by the World Bank as upper-middle or high-income countries.
The deal was signed on July 30, according to an SEC filing by Novavax.
On Tuesday, Novavax reported that its experimental COVID-19 vaccine produced high levels of antibodies against the novel coronavirus in a small, early-stage clinical trial, and that it could start a large pivotal Phase III trial as soon as late September.
AstraZeneca in deal with China's Kangtai Bio
AstraZeneca PLC has signed an exclusive framework agreement with China's Shenzhen Kangtai Biological Products to supply its COVID-19 vaccine candidate in mainland China, the British pharmaceutical giant said on Thursday.
To meet market demand in China, Shenzhen Kangtai is obliged to make sure it has an annual production capacity of at least 100 million doses of the experimental shot AZD1222, which AstraZeneca co-developed with researchers at Oxford University, by the end of this year, and a capacity of at least 200 million doses by the end of next year, AstraZeneca said in a statement on the Chinese social media site WeChat.
The two companies will also explore the possiblity of cooperation on the vaccine candidate in other markets, AstraZeneca said.
Zydus Cadila's vaccine candidate found safe in early-stage human trial
BENGALURU: India's Zydus Cadila said on Wednesday its COVID-19 vaccine candidate was found to be safe and well-tolerated in an early-stage human trial.
The company will now start a mid-stage trial of the vaccine candidate, ZyCoV-D, in over 1,000 healthy adult volunteers from Thursday to test its effectiveness, it said in a regulatory filing.
Zydus plans to complete late-stage trials for ZyCoV-D by February or March and could produce up to 100 million doses a year initially, the company's chairman told Reuters last month.
The safety of the vaccine candidate in volunteers enrolled in the early-stage trial, who were administered doses since July 15, was endorsed by an independent data safety monitoring board.
Zydus is also among the several Indian generic drugmakers that have licensing agreements with U.S.-based Gilead Sciences to produce remdesivir, the antiviral that has been approved in the country as an emergency treatment to fight the coronavirus outbreak.
India reported a daily jump of more than 50,000 coronavirus infections for the seventh straight day on Wednesday. Total cases now stand at 1.91 million, the third biggest, after the United States and Brazil.
Global recovery could be faster if vaccine made available to all: WHO chief
Economic recovery around the world could come faster if any COVID-19 vaccine is made available to all as a public good, World Health Organization Director General Tedros Adhanom Ghebreyesus.
He was speaking in an online panel discussion on Thursday with members of the Aspen Security Forum in the United States moderated by the NBC network.
"Sharing vaccines or sharing other tools actually helps the world to recover together. The economic recovery can be faster and the damage from COVID-19 could be less," Tedros said.
"Vaccine nationalism is not good, it will not help us," he said in an allusion to the competitive scramble of nations and pharmaceutical researchers to come up with an effective vaccine and order as many doses as possible in advance.
Tedros had said on Monday that while the coronavirus was the biggest health emergency since the early 20th century, the international race for a vaccine was also "unprecedented".
"We must seize this moment to come together in national unity and global solidarity to control COVID-19," he told Thursday's forum. "No country will be safe until we are all safe." WHO emergencies director Michael Ryan, asked about a proposed Russian vaccine, told the panel that trial data was needed to ensure any vaccines are safe and effective.
Ryan also said authorities should be able to demonstrate the efficacy of a coronavirus vaccine via traditional clinical trials rather than "human challenge" studies.
He was referring to the intentional exposure of vaccinated volunteers to a virus to see whether the vaccine works.
Vaccine before November 3 US elections: Trump
US President Donald Trump said on Thursday it was possible the United States would have a coronavirus vaccine before the Nov. 3 election — a more optimistic forecast than timing put forth by his own White House health experts.
Trump has accused the WHO of becoming a puppet for China — where the coronavirus outbreak first surfaced late last year — during the COVID-19 pandemic and given notice that the United States will quit the agency in a year's time.
The United States is the biggest overall donor to the WHO and had contributed more than $800 million by the end of 2019 for the 2018-19 biennial funding period.
But Tedros, who has denied that the WHO answers to China or any other country, told the panel that the main damage from the Trump administration's move to exit the Geneva-based agency would not be the loss of funding.
"The problem is not about the money, it's not the financing..., it's actually the relationship with the U.S. That is more important for the WHO - the void, not the financial. And we hope then U.S. will reconsider its position," he said.








