COVID-19: Do intranasal vaccines work better?
2 Indian pharma giants pursue late-stage intranasal vaccine clinical trials
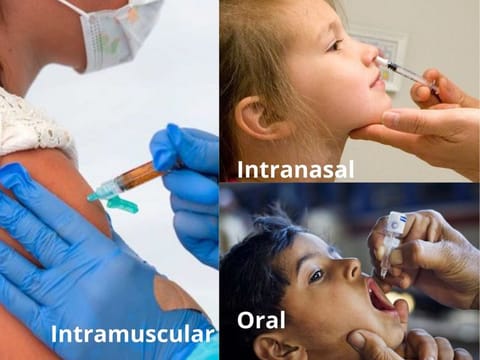
DUBAI: Could an intranasal vaccine be more effective than instramuscular shots trained against COVID-19? This question is currently foremost in the minds of a number of vaccine developers. Recently, a research team did a comparative study in mice, with results suggesting that intranasal vaccines were more effective in conferring immunity to SARS-CoV-2 than intramuscular shots.
How are vaccines administered?
Vaccines are delivered via different routes. The polio vaccine, for example, is an oral drop. Most vaccines are administered through the muscles (intramuscular) or the tissue just between the skin and the muscles (subcutaneous).
A relatively new route pharmaceutical researchers want to take is the nasal route, in which the vaccine is sprayed into the nostrils and inhaled. Researchers are working towards getting this delivery model. The nasal flu vaccine is a classic example of a nasal shot.
What are the advantages of intranasal vaccine?
One, it helps overcome potential difficulties with distributing the vaccine and the high cost of vaccination. The use of intranasal vaccines against coronavirus are also expected to help solve the possible shortage of trained personnel to administer the doses.
HOW INTRANASAL VACCINES WORK
Intranasal vaccines are administered by spraying/squirting the solution into the nostrils, instead of injected it in the arm.
The study, published in the journal Cell as a pre-proof, has shown that an intranasal administration of an experimental COVID-19 vaccine worked better in animal models, than the intramuscular route. In the new study, the researchers found that the nasal delivery created a strong immune response throughout the body. But it was found particularly effective in the nose and respiratory tract, preventing the infection from taking hold in the body. Based on available literature, most COVID-19 vaccines in development are planned to be intramuscular shots.
Who developed the vaccines?
In India, two vaccine makers — Serum Institute of India (SII) and Bharat Biotech — have announced the development of intranasal vaccines. Bharat Biotech has reportedly collaborated with the Washington University School of Medicine in St Louis, Missouri, to develop an intranasal shot against COVID-19. Both Indian firms are expected to pursue late-stage clinical trials of intranasal COVID-19 vaccines in the coming months once they receive regulatory approval, India's health minister said on Sunday.
The University of Alabama at Birmingham (UAB) also stated they are running pre-clinical trials of intranasal COVID-19 vaccine candidate, called AdCOVID. It was developed by US-listed pharma company Altimmune. On October 12, the company reported that comprehensive pre-clinical evaluation shows a single-dose intranasal administration of AdCOVID stimulates 3 key immune components: serum-neutralising antibody, T cell responses, and mucosal immunity in the respiratory tract.
AdCOVID
AdCOVID is based on the Altimmune's adenovirus-based intranasal vaccine platform and expresses the receptor binding domain (RBD) of the SARS-CoV-2 spike protein. The RBD is essential for viral infection and the majority of neutralising antibodies from people that have recovered from COVID-19 bind to the RBD, highlighting the importance of the RBD in controlling infection. AdCOVID is designed to work with a single dose, trigger a broad activation of immune response, and the "ability to ship and store the vaccine conveniently and inexpensively," said the company.
What are the findings of the AdCOVID intranasal vaccine study?
In a BioRxiv.org (pre-print) publication, the authors presented data which indicate strong activation of adaptive immune system following a single intranasal dose of AdCOVID. These data, gleaned from tests on two strains of mice, show that AdCOVID stimulated strong immune responses.
Serum-neutralising immunity: AdCOVID elicited a median serum neutralization titer against wild-type SARS-CoV-2 virus of up to 1:563 one-month post-vaccination in a 50% focus reduction neutralisation test (FRNT). This level of neutralising activity was at least 3-fold higher than the minimum titer recommended by the FDA for convalescent plasma used in the treatment of COVID-19.
T cell immunity: AdCOVID stimulated both CD4+ and CD8+ antigen-specific T cell responses following a single intranasal vaccination. The response was focused in the lungs of the vaccinated mice and was biased toward CD8+ T cells. A significant fraction of the CD8+ T cells in the lung were found to be non-circulating tissue-resident memory (Trm) T cells, which have been shown to play a front-line role in fighting respiratory viral infections.
Mucosal immunity: Nasal mucosal immunity is a local type of immunity that has the potential to stop both infection and transmission of the virus. Altimmune claimed only an intranasal vaccine can activate this important type of immunity.
How many intranasal doses are planned by Indian vaccine makers?
Bharat Biotech, based in the Indian city of Hyderabad, has stated it will be manufacturing up to a billion doses of a single-dose intranasal COVID-19 vaccine. Dr Harsh Vardhan said the late-stage trial generally involves thousands of participants, sometimes 30,000 to 40,000. Of the vaccines currently in Phase 3 trials, all are administered by injection, according to the World Health Organization (WHO).
On Saturday, India's Dr Reddy's Laboratories Ltd and the Russian Direct Investment Fund (RDIF) said they have received renewed approval to conduct late-stage clinical trials in India of the Russian COVID-19 vaccine. India's COVID-19 infections rose by another 61,871 over the past 24 hours, data released on October 19 showed, with 115,000 deaths reported by the country's health authorities.
The Indian Council of Medical Research (ICMR) said it will revisit its protocol for COVID-19 treatment after the WHO found some of the commonly used drugs, including remdesivir, had little or no impact on a patient's chances of surviving, local media reported earlier on Sunday.
Also Read: Vaccine for COVID-19 close at hand?
Also Read: Facebook bans ads discouraging vaccines
When was intranasal shot first approved?
In 2003, the US Food and Drug Administration approved "FluMist", a nasal spray vaccine against the Influenza virus. The approval came following 40 years of research by Arab-American researcher Professor Hunein “John” Maassab of the University of Michigan (UM). FluMist wsa approved for use by healthy people ages 5-49, is a cold-adapted, "live-attenuated" (virus is still alive, but weakened), trivalent influenza virus vaccine.
It was the first flu vaccine delivered as a nasal mist to be commercially available in the US. Other vaccine makers soon followed the nasal route.
How many nasal spray vaccines are used each year?
In 2009, the CDC rolled out the first H1N1 (swine flu) vaccines in US in the form of a nasal spray, initially with 3.4 million doses of MedImmune (AstraZeneca). It was later bumped up to about 20 million doses a week. It was given to people in America for free. That year alone, the US has ordered 195 million doses of H1N1 swine flu vaccine from five pharmaceutical companies.







