Also In This Package
Mumbai lockdown: Shaan and other Bollywood stars react
Meet the 10 crypto billionaires on the 'Forbes' list
In photos: Janhvi Kapoor holidays in Maldives
Photos: The life and times of Britain's Prince Philip
Richest Filipinos: Wealth grows during COVID-19
Look: On Dubai’s Palm, it’s The View that matters
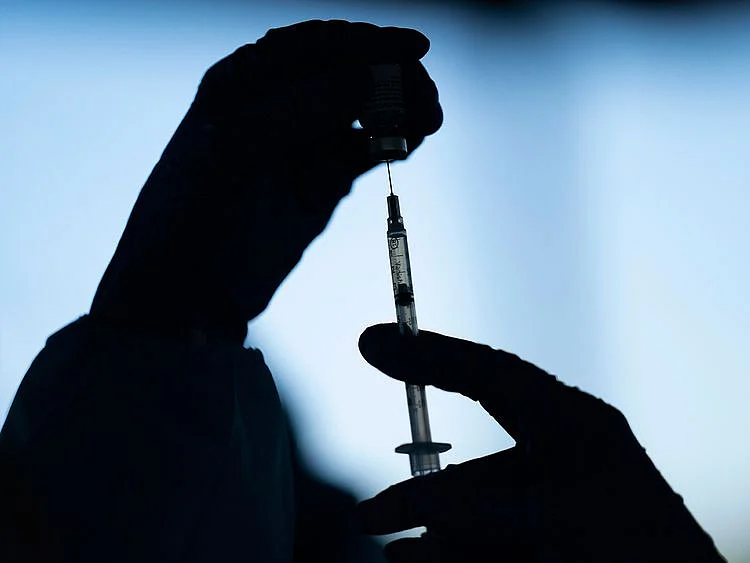
Washington: Pfizer-BioNTech asked for authorization on Friday to use their COVID-19 vaccine on 12-15 year olds in the United States.
The companies said in a statement that they plan to make similar requests of other regulatory authorities worldwide in coming days.
Sign up for the Daily Briefing
Get the latest news and updates straight to your inbox
Network Links
GN StoreAbout Gulf NewsTerms & ConditionsReach by GNSitemapContact usPrivacy PolicyGN FocusHave your sayWork with usAdvertise with usGulf News epaperPrinting Services
Download our app
© Al Nisr Publishing LLC 2026. All rights reserved.