Zolgensma, the Dh8 million SMA drug: When high cost makes it difficult to go for treatment
Zolgensma: Sometimes the high cost makes it difficult to go for treatment
What is the value of human life? It is priceless; too sacred to put a value to it, most would say. In general, we have been conditioned to resist the idea of putting a price on human life but the concept is not at all alien to us. The issue becomes clearer if the question is put differently. How much are you ready to spend on saving one life? Uncomfortable as it might sound, one is often faced with situations when the cost of treatment makes it difficult to make the choice. Treatment involving Dh7.9m Zolgensma, touted as the costliest drug, is one such thing. Let us take a look at the drug and the condition it treats.
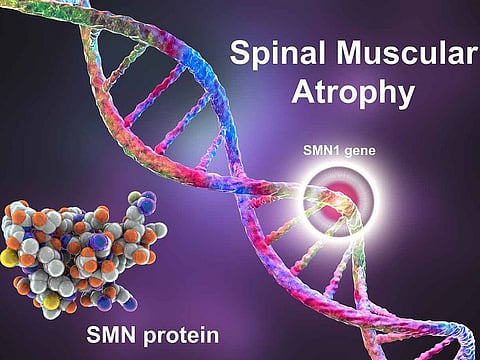
Spinal Muscular Atrophy and Zolgensma
When parents of Shivaraj Daware, an Indian toddler from Nashik, Maharashtra, were told that Zolgensma could help save their toddler son diagnosed with debilitating Spinal Muscular Atrophy or SMA, they were in no position to even digest the information.
The staggering cost of Rs160 million (Dh7.9m) for the experimental drug is far beyond the means of most people in India, where health insurance is inadequate and not available to all. And for the Dawares, whose main income was from a photocopy shop, it was beyond their wildest hopes.
It was then that Dr Brajesh Udani of Hinduja Hospital told them that they could get the potentially life-saving drug through a lucky draw as part of the US firm Novartis’s clinical trials. Zolgensma, or onasemnogene abeparvovec-xioi, was approved in 2019 in the US. The draw was held on December 25, 2020, and Shivaraj became India’s first patient to be administered the drug on January 19, 2021.
Novartis says more than 1,200 children have been treated with Zolgensma so far and claims that data has shown life-changing benefits for the patients who have undergone the single-dose gene therapy.
The case of Shivaraj Daware received huge media coverage in India which resulted in considerable interest in Zolgensma and Novartis. Soon, several cases that required the use of Zolgensma began to crop up.
In early 2021, the case of Mumbai-born Teera Kamat set a trend in another direction. With this, crowdfunding entered the scene as a viable option to raise such huge sums for treatment in India. Suddenly, despite the prohibitive cost, treatment became possible albeit with a generous dose of help from social media and charity funding.
Mihir and Priyanka Kamat, parents of 5-month old Teera, managed to raise Rs160 million with the help of some 100,000 donors across the world. The case attracted the attention of Prime Minister Modi who intervened to give Rs60 million tax exemption for importing the costly drug to India.
Another case of SMA patient that garnered considerable public interest was that of Imran, son of Arif in Kerala's Kozhikode. Crowdfunding efforts raised a huge amount for him, but unfortunately, the child died before he was given the treatment.
What is SMA?
Spinal muscular atrophy (SMA) is a genetic neuromuscular disease that causes certain muscles to become weak and waste away. SMA is a progressive disorder – it gets worse over time - and affects motor neurons in the spinal cord that control muscles. It affects around one in 10,000 live births.
What are neurons? Neurons are the fundamental units of the brain and the nervous system. These cells are responsible for receiving sensory inputs from the external world and send out motor commands to our muscles. There are three different types of neurons: sensor, motor and interneurons.
Most of the nerve cells that control the muscles are located in the spinal cord. The muscles become smaller and waste away when they do not get stimulation from the nerve cells. It affects the central nervous system, peripheral nervous system, and voluntary muscle movement.
SMA causes progressive loss of motor neurons resulting in gradual loss of muscle tone, muscle weakness and atrophy. Though a rare disorder, it is the most common genetic cause of mortality in infants if untreated.
The age at which SMA symptoms begin roughly correlates with the degree to which motor function is affected, according to Muscular Dystrophy Association.
The earlier the age of onset, the greater the impact on motor function. Children who display symptoms at birth or in infancy typically have the lowest level of functioning.
When things go wrong with neurons
SMA is caused by a mutation of the Survival Motor Neuron1 gene, which is responsible for the protection and survival of motor neuron protein which in turn maintains the healthy functioning of the motor neurons.
It is a paralytic infantile-onset disorder caused by low SMN protein. Untreated, most patients with SMA die before 2 years of age.
The disorder has been classified into five types, with different levels of severity.
There are five different SMA types
What is Zolgensma?
It is a one-time-only prescription gene therapy for children aged less than two years with spinal muscular atrophy (SMA), a genetic disorder that causes paralysis, muscle weakness, progressive loss of movement, and difficulty breathing and swallowing.
The most expensive drug in the world at nearly $2.5m a dose, Zolgensma is given as an infusion into a vein. The active ingredient onasemnogene abeparvovec enters the motor neuron cells (nerves) and restores the missing or non-working SMN1 gene with a new copy. The new SMN1 produces proteins essential for nerve function and controlling muscle movement.
Why is Zolgensma important in the treatment of SMA?
Zolgensma, produced by Swiss pharma company Novartis, is one of three approved treatments available for SMA. It is reported to have helped babies breathe without a ventilator and move around without much help. But it can only be administered to patients less than two years old.
The gene therapy is a one-time treatment, while Spinraza, an alternative to Zolgensma, has to be taken three or four times a year for life. That too through a lumbar puncture.
Research has been underway for its use in patients between 2 and 18 years. Now, with the US Food and Drug Administration lifting its restrictions, Novartis will launch a new Phase 3 trial to study the use of Zolgensma for Type 2 SMA patients aged between 2 and 18, who can sit, but never walked.
Spinraza; Close contender
The only other drug approved by FDA for treating is Spinraza by Biogen, another US biotechnology giant. Spinraza has a price of $750,000 for the first year of treatment, but that doesn’t make it cheaper compared to Zolgensma. Because unlike Zolgensma, which is a one-time drug, Spinraza has recurring costs. From the second year onwards, its price drops to $375,000 per year and continues so for the remainder of the patients’ lives, making it effectively costlier than Zolgensma.
What is the life of an SMA patient without Zolgensma?
Babies born with severe Type 1 SMA (Werdnig-Hoffmann disease, the most common form) have only two years of life expectancy. These infants usually fail to reach developmental milestones such as rolling, sitting or crawling.
Long-term clinical data showed that a single dose of Zolgensma extends the survival of Type 1 SMA patients and prevents motor neuron loss for several years. Without treatment, around 68% of children die before their second birthday, and 82% die before their fourth, a report in SMA News Today said.
Type 0 is the most severe form of the disease, and the onset takes place before birth. Babies with Type 0 SMA “show severe muscle weakness, decreased muscle tone, and are unable to develop age-appropriate motor skills. Severe respiratory problems are common, and few live longer than six months after birth”, SMA News Today reported.
Milder forms of SMA like Type 2 (Dubowitz disease) and Type 3 (Kugelberg-Welander disease) appear later in childhood, while Type 4 (adult-onset SMA) is seen in young adults who live longer and can walk unaided until the age of 60. People with types 2, 3 and 4 generally live into adulthood with proper care and breathing support.
Michaela Hollywood, who suffers from Type 2 SMA, works to raise awareness of the disorder. She was included among 100 of the world’s most inspirational women of 2015 by the BBC.
What are the SMA treatments?
Spinraza and Evrysdi are the two other SMA treatments approved by the FDA. While Spinaraza can be used to treat patients of all ages and types of SMA, Evrysdi has been approved for the treatment of adults and children aged over two months.
Biogen’s Spinraza (generic name nusinersen) was the first accessible treatment for SMA. Unlike Zolgensma, which targets SMN1, Spinraza is a tiny piece of DNA that works on the SMN2 gene to create the protein the body needs.
The drug has to be injected into the spinal column by a lumbar puncture to reach the motor neurons. This can be challenging for older children and adults as they are likely to have a spinal curvature. It also needs to be administered up to six injections in the first year of treatment, and at least three each year after that, a report in The Conversation said.
“But neither of these drugs [Zolgensma and Spinraza] can be regarded as a complete cure, particularly for patients who have already developed significant symptoms. The timing of treatment for patients is critical, as the human body cannot replace motor neurons once they are lost. Ideally, treatment should be carried out before symptoms are developed,” Judith Sleeman, senior lecturer in cell biology at the University of St Andrews, United Kingdom, wrote in The Conversation.
Evrysdi (generic name risdiplam) from Roche is not gene therapy, so patients must be dosed every day. The oral therapy, used in treating adults and children aged over two months, works by preventing the SMN2 genes from disrupting protein production.
Some researchers think that a combination of SMN and non-SMN treatments could be more effective in treating SMA. It means that patients will take two drugs together or take one medicine at one stage of the disease and another medicine at a different stage.
Can I prevent spinal muscular atrophy?
The answer is more complicated than a simple “yes” or “no”. SMA is an inherited disease. If you or your spouse carries the mutated gene that causes SMA, a genetic counsellor can explain the chances of your child having SMA or being a carrier.
Steps can be taken before pregnancy to lower the risk of passing on SMA. This can be done through a procedure called “pre-implantation genetic diagnosis” (PGD), which helps identify embryos that don’t have the mutated gene.
Your doctor implants healthy embryos during in-vitro fertilisation (IVF). PGD ensures your child will have two healthy SMN1 genes and not get SMA.
Why screen for SMA?
When screening is done before or during pregnancy, it allows parents to find out the chances of having a child with a genetic disorder.
How is SMA detected, or diagnosed?
In general, a carrier screening test is performed to tell whether you carry a change in a gene for certain genetic disorders. With SMA, detection is done through SMA-carrier screening.
This procedure helps determine if a couple is at increased risk for having a child with SMA.
Some SMA symptoms resemble those resulting from neuromuscular disorders like muscular dystrophy. To find out what triggers symptoms, a physical check is done and medical history is taken.
One or more of these tests may have to be done to diagnose SMA:
Blood test:
An enzyme and protein blood test can check for high levels of creatine kinase. Deteriorating muscles release this enzyme into the bloodstream.
Genetic test:
This blood test identifies problems with the SMN1 gene. As a diagnostic tool, a genetic test is 95% effective at finding the altered SMN1 gene. Some states test for SMA as part of routine newborn screenings.
Nerve conduction test:
An electromyogram (EMG) measures the electrical activity of nerves, muscles and nerves.
Muscle biopsy:
Rarely, a physician may perform a muscle biopsy. This procedure involves removing a small amount of muscle tissue and sending it to a lab for examination. A biopsy can show atrophy, or loss of muscle.
SMA in family
- You could be a carrier of SMA even if no one in your family has SMA and even if you already have children without SMA. - If you have a family member who has SMA, it means that your risk of being a carrier is increased. If both partners are carriers, and the couple is thinking of becoming pregnant, there are options available for them. [Source: clevelandclinic.org]
What is pre-natal screening for SMA; how does it help?
Tests can be performed to determine if the foetus has inherited two copies of the SMA gene mutation. In general, there are two prenatal screening for SMA:
(a) Chorionic villus sampling (CVS), performed at 10 to 14 weeks.
(b) Amniocentesis, done at 16 to 20 weeks.
Age up to 2 years is crucial for an SMA patient. Why?
Symptoms of SMA often begin at birth or within the first six months of life. Some babies with SMA are "floppy" — unable to roll or sit at the expected age. An older child may fall more often than others.
To better understand the age factor, it’s best to know the 4 SMA types. The first three (Types 0, 1 and 2) are usually seen or diagnosed before the age of 2. Type 0, the rarest and most severe, is found before a child is born. Type 1, known as Werdnig-Hoffman (infantile-onset SMA), is a very severe type and may be present at birth. Most children with type 1 SMA die before their second birthday.
Type 2, also called Dubowitz disease (intermediate SMA), is seen in children from age 6 months to 18 months.
Children with Type 2 SMA have overall muscle weakness. This type tends to affect the lower limbs: they may be able to sit up but can't walk. They may need braces, a walker, or a wheelchair. They can often live into adulthood.
In Type 3 SMA, also called Kugelberg-Welander (mild SMA), symptoms may appear in children between ages 2 and 17 years. Symptoms include clumsiness, trouble walking, mild muscle weakness and delayed motor development. Children with Type 3 can live long into their adult years.
Who makes Zolgensma?
Zolgensma, sold by Novartis, is a one-time treatment for spinal muscular atrophy (SMA).
Why is Zolgensma costly?
Zolgensma belongs to a new class of highly specialised drugs — gene therapy. It is the second and most effective drug for spinal muscular atrophy (SMA). The reason for its outrageously high cost is its rather small market size and its potential to save lives — SMA being a rare disorder. Novartis bought Zolgensma in 2018 with its $8.7 billion purchase of AveXis. Reuters, quoting company data, reported that Zolgensma sales were $291 million in Q3 2020 and $254 million in Q4 2020.
Putting a price tag on human life
How much are you ready to spend on saving one life? Whose life, is the question most of us would ask, because, we know,
1. There’s a price tag on human life, and
2. it is relative, the closest being the priciest.
And we do it consciously and unconsciously on all fronts. It also depends on who you ask.
For a statistician, a human life is just one out of roughly 7.9billion of our species making the earth fast uninhabitable. Which is absolutely next to nothing, unless it is his or her significant other or immediate family or friends. However, for an economist or insurance agent or industrial safety evaluator or policymaker, it is a hard reality. Something they have to process frequently in the course of their professional activity. So after all what is the value of a human life?
Value of life
Contrary to what many think, value of life is not a strange concept despite the general feeling. There is a monetary value attributed to human life and it is quantified based on the benefits of avoiding a fatality and the costs implied in averting it.
And do you want a solid figure? One human life is currently worth approximately $10 million, according to Kip Viscusi, Professor of Law, and Economics at Vanderbilt University. He calls it a statistical life and it is the value the US government agencies reportedly uses when making money vs life decisions.
Insurance cover for Zolgensma in UAE
Insurance providers are most often the only saviours for people hard hit for cash to buy medicines. For the most expensive medicine in the world, it is a different scenario altogether: only a few health insurance providers cover Zolgensma.
Explaining why it is not offered by insurance companies in the UAE and other parts of the world, Avinash Babur, CEO of UAE-based InsuranceMarket.ae, said: "It is a relatively new treatment and as per reports there is a lack of conclusive evidence of its effectiveness".
Also, the high cost ($2 million for a one-time infusion lasting one hour) are the prime reasons for insurance companies not only in the UAE but around the globe for not offering the medicine, he said.
After extensive clinical trials, Zolgensma was approved in the US in May 2019. The US Food and Drug Administration approved the medicine for IV delivery in patients with all types of SMA who are under two years of age.
Zolgensma has been approved by more than 38 countries for use, but that’s not the case with insurance providers.
In some countries, including the US, selective insurance providers support SMA treatment. The pharmaceutical giant Novartis recently said 20 commercial insurance plans and four Medicaid plans have policies in place on coverage of its gene therapy Zolgensma.
The rationale behind it all, according to Babur, is: "Insurance companies need to consider the effects to their balance sheets in offering to cover the costs of such treatments. Just as drug developers and manufacturers need to pass their costs on, so do insurance companies: resulting in higher premiums across the board for health insurance customers."
How insurance works
Insurance companies work based on a 'pooling system', the claims of a few funded by many, said Babur. This means that the cost is shared by all and it is not just the patients who make a health insurance claim who are subjected to a price increase, explained the CEO of InsuranceMarket.ae.
"To the policyholder, their case is an isolated one, but in agreeing to cover one claim, a precedent is set, that is to say 'to offer to one means to offer all'. In the case of high-value treatments, the cost of a single claim could run into several million and for many insurance companies, even one claim may be too much to financially cover, let alone further claims in addition. Their business would be unsustainable and this would affect other policyholders’ claims."
Gene therapies are still relatively embryonic and evolving, treatments and drugs falling in this category remain very costly. Medicine manufacturers need to recoup their extensive research and development costs (including clinical trials) and the price point of their products will reflect this. Similarly, as pioneering products, their efficacy is not well known. These are just two reasons why treatments generally fall within the list of exclusions under an insurance policy, Babur said.
Crowdfunding for Zolgensma
When insurance providers and drug manufacturers turn down the needy, they turn to the very last resort: crowdfunding. When a drug costs millions, apart from sourcing cost and tax, the cash-strapped patients and their parents seek the help of fellow human beings. The money is either collected through a bank account and the message is either spread through the internet or media outlets.
Some success stories of crowdfunding for Zolgensma
More than Rs470 million (Dh23.48 million) was raised to buy Zolgensma for an 18-month-old baby suffering from spinal muscular atrophy in the south Indian state of Kerala using crowdfunding. The treatment cost for the medicine is around 180 million and the parents of the child said the rest of the money would be utilised for other SMA patients.
ImpactGuru.com, India’s leading healthcare financing platform for patients, has raised Rs 160 million (Dh 8.95 million) to cover the cost of Zolgensma for a 3-month-old baby diagnosed with Spinal Muscular Atrophy Type 1 in just 40 days.
Parents of a two-year-old baby in Singapore launched a crowdfunding campaign with Ray Of Hope to raise $2.868 million for their son, who was diagnosed with Type 2 Spinal Muscular Atrophy, to get a one-time-only gene therapy treatment Zolgensma.
Other parents in countries including Belgium, Slovenia, Greece, Hungary, Cyprus and Estonia have taken similar measures to access Zolgensma for their children, according to Politico.
Portugal, France and Germany have agreed to pay under provisional agreements allowing patients access to drugs that treat life-threatening diseases before they're approved in Europe, according to Politico. Other countries, such as Estonia, have refused to reimburse Zolgensma before it hits the market, arguing it's unaffordable.



